LATISSE® Minneapolis

Are your eyelashes thinner, shorter, and paler than they used to be? Are there lashes missing? If so, you may have a condition called hypotrichosis. This is easily treatable with the help of convenient and long-lasting Latisse®.
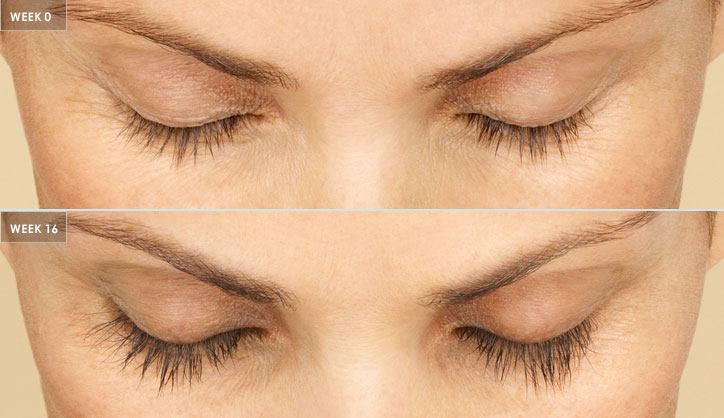
With FDA-approved Latisse® eyelash-regrowth medication, men and women can restore full, dark, thick lashes. Patients apply a single drop to each upper eyelid once per day for a total of sixteen weeks. Typically, results begin to be visible after four weeks of daily application.
To learn more about prescription Latisse®, contact our office and arrange a consultation with Dr. Lokeh.
What is LATISSE®?
LATISSE® (bimatoprost ophthalmic solution) 0.03% is indicated to treat hypotrichosis of the eyelashes by increasing their
growth, including length, thickness, and darkness.
How does LATISSE® work?
Eyelash growth is cyclical.1-3 The normal eyelash growth cycle involves 4 phases—anagen, catagen, telogen, and exogen—and occurs over several months. LATISSE® is believed to prolong the active growth phase—or anagen phase.
courtesy of Latisse
It’s important for patients to understand that LATISSE® solution works gradually. While they may start seeing longer lashes after 4 weeks, they must use LATISSE® daily for 16 weeks for maximum results.4,5 They should not reduce or stop daily application of LATISSE® when they first notice results.
What are some common side effects?
ncreased iris pigmentation has occurred when bimatoprost solution was administered. Patients should be advised about the potential for increased brown iris pigmentation, which is likely to be permanent.
Bimatoprost has been reported to cause pigment changes (darkening) to periorbital pigmented tissues and eyelashes.
There is the potential for hair growth to occur in areas where LATISSE® solution comes in repeated contact with skin surfaces. Apply LATISSE® only to the skin of the upper eyelid margin at the base of the eyelashes.
LATISSE® solution should be used with caution in patients with active intraocular inflammation (eg, uveitis) because the inflammation may be exacerbated. LATISSE® contains benzalkonium chloride, which may be absorbed by soft contact lenses. Contact lenses should be removed prior to application of solution and may be reinserted 15 minutes following its administration.
Adverse Reactions: The most frequently reported adverse events were eye pruritus, conjunctival hyperemia, skin hyperpigmentation, ocular irritation, dry eye symptoms, and erythema of the eyelid. These events occurred in less than 4% of patients.